


How full of the creative genius is the air in which these are generated! I should hardly admire more if real stars fell and lodged on my coat. Nature is full of genius, full of divinity: so that not a snowflake escapes its fashioning hand.
-Henry David Thoreau, Journal, 1856
The wonder of how a snowflake forms is one that incites curiosity from both children and adults alike. All of us, at one time or another, have watched a snowflake land and taken an up-close view of the intricate latticework that embodies the snowflake. The form of each snowflake is a remarkable piece of artwork that nature provides us each winter. Even more remarkable, is the fact that nature never repeats the same design twice, as each snowflake is unique. The intricacy of each snowflake can be understood by digging into how a snowflake first forms, and then looking into what factors along the way contribute to the final result.
First we must start by journeying up into the clouds where it all begins. Clouds are typically composed of a mix of ice crystals and supercooled water droplets (water that is colder then 32F, but doesn't freeze). Amazing as it sounds, water droplets tend to remain in the liquid form all the way down to temperatures of -38F. This is because in order for water to freeze at a temperature greater than -38F, it needs an ice nucleus. Without an ice nucleus, the water droplets will only be able to freeze if they get colder than -38F. Therefore, most of the time, ice nuclei are needed. Some examples of particulates that serve as ice nuclei are desert dust, volcanic ash, pollen, and even a certain strand of leaf bacteria.
When the supercooled droplets freeze, they take on an initial form that is driven by the molecular structure of water. Water molecules inherently freeze in the form of a hexagonal structure (a six-sided shape). This initial shape is driven by the hydrogen bonding that exists within a water molecule. At this point, the ice crystal remains light enough that it remains suspended in the cloud. While suspended in the cloud, other supercooled droplets freeze onto the initial ice crystal. This allows the crystal to steadily grow in size. The additional supercooled water droplets that the ice crystal comes in contact with will tend to freeze on the outside edges of the initial hexagonal form, resulting in the growth of 6 branches that extend outward from the center of the snowflake. The image below depicts this process starting at the initial ice crystal formation, followed by the growth that occurs as surrounding supercooled water droplets continue to freeze onto it.
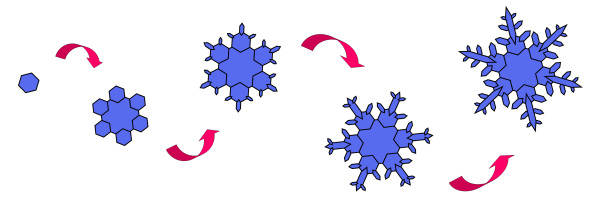
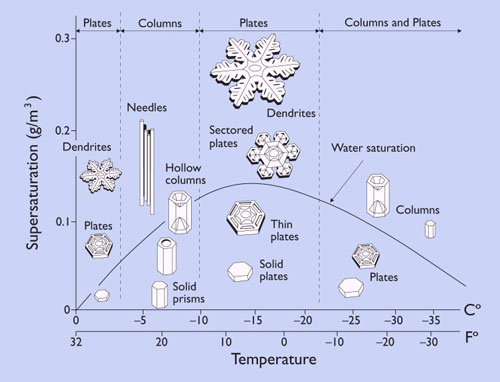
As the snowflake travels through different portions of the cloud, it continues to grow, encountering contrasting temperatures and humidity levels along the way. The specific temperature and humidity level present in the cloud layer correlates to the type of form the snowflake takes on. Snowflakes that form in cold and dry environments tend to take on a simpler form, as less moisture results in less intricate crystal growth. Conversely, when snowflakes grow in warmer and increasingly moist environments, their form tends to take on the intricate dendrite form (typical 6 branched snowflake) that we are all familiar with. The image below provides the varied snowflake shapes that occur when ice crystals grow in environments of differing moisture and temperature combinations (moisture level is the vertical or Y axis while temperature is the horizontal or X axis).
Once the snow crystal has grown heavy enough, it can no longer be supported by the weak updrafts present in the clouds (slow rising air currents). At this point, it will begin to fall from the cloud and begin its trek down to the surface. Along its way down, the snowflake's shape will undergo further modification due to collisions with other snowflakes. When snowflakes fall into a warmer environment (temperatures close to 32F), the edges of the snowflake will partially melt. When this happens, other snowflakes that come into contact with it will tend to stick to it. This process of snowflakes sticking together to form larger snowflakes is called aggregation. This is common when temperatures are near the freezing mark as the snowflake gets closer to the surface. Conversely, when snowflakes travel down into a very cold and well below freezing environment, the snowflakes take on a more brittle character and will fracture and break apart when they collide with one another. The last factor that can impact the eventual shape of the snowflake is wind. When high winds are encountered during the snowflakes descent to the ground, crystal fracturing will occur for both wet as well as the dry, brittle variety of snowflakes.
With all the variables that occur from the time an ice crystal first forms, to when the finished product of a snowflake reaches the ground, it is not hard to grasp the concept of no two snowflakes ever being alike. Each individual snowflake will travel in at least a slightly different path from each other, meaning it will travel through varied temperature and humidity levels at differing times from one another. The different temperatures and humidity levels that the ice crystal travels through, and how long it is subjected to each environment, directly impacts the type of form each crystal will take on. The end result of it all is one of nature's great pieces of artwork, lending to the sense of wonder that the viewing of a snowflake up-close creates.